by Bo G. Malmström and Bertil Andersson*
1. Introduction
1.1 Chemistry at the borders to physics and biology
The turn of the century 1900 was also a turning point in the history of chemistry. Consequently, a survey of the Nobel Prizes in Chemistry during this century will provide an analysis of important trends in the development of this branch of the Natural Sciences, and this is the aim of the present essay. Chemistry has a position in the center of the sciences, bordering onto physics, which provides its theoretical foundation, on one side, and onto biology on the other, living organisms being the most complex of all chemical systems. Thus, the fact that chemistry flourished during the beginning of the 20th century is intimately connected with fundamental developments in physics.
In 1897 Sir Joseph John Thomson of Cambridge announced his discovery of the electron, for which he was awarded the Nobel Prize for Physics in 1906. He found that these negatively charged ‘corpuscles’, as he called them, have a mass 1000 times smaller than the hydrogen atom. Thomson’s discovery had, of course, important implications for chemistry, as it showed that the atom is not an indivisible building block of chemical compounds, but it took a number of years before this led to developments of direct relevance to chemistry. In 1911 Ernest Rutherford, who had worked in Thomson’s laboratory in the 1890s, formulated an atomic model, according to which the positively charged atomic nucleus carries most of the mass of the atom but occupies a very small part of its volume.
This is instead created by a cloud of electrons circling around the nucleus. Rutherford received the Nobel Prize for Chemistry already in 1908 for his work on radioactivity (see Section 2).
It was soon realized that in Rutherford’s atomic model the stability of atoms was at variance with the laws of classical physics, since the electrons would lose energy in the form of electromagnetic radiation and eventually fall into the nucleus. Niels Bohr from Copenhagen understood that an important clue to the solution of this problem could be found in the distinct lines observed in the spectra of atoms, the regularities of which had been discovered in 1890 by the physics professor Johannes (Janne) Rydberg at Lund University. Consequently, Bohr formulated in 1913 an alternative atomic model, in which only certain circular orbits of the electrons are allowed. In this model light is emitted (or absorbed), when an electron makes a transition from one orbit to another. Bohr received the Nobel Prize for Physics in 1922 for his work on the structure of atoms.
Another step in the application of the electronic structure of atoms to chemistry was taken in 1916, when Gilbert Newton Lewis suggested that strong (covalent) bonds between atoms involve a sharing of two electrons between these atoms (electron-pair bond). Lewis also contributed fundamental work in chemical thermodynamics, and his brilliant textbook, Thermodynamics (1923), written together with Merle Randall, is counted as one of the masterworks in the chemical literature. Much to the surprise of the chemical community, Lewis never received a Nobel Prize.
Even if the contributions just described were made a decade or more after Thomson’s discovery, much important work in the borderland between physics and chemistry was published in the 1890s, and this was naturally given a strong consideration by the first Nobel Committee for Chemistry (see Section 2). In fact, three of the Laureates during the first decade, Jacobus Henricus van’t Hoff, Svante Arrhenius and Wilhelm Ostwald, are generally regarded as the founders of a new branch of chemistry, physical chemistry. Fundamental work had, however, also been done in more traditional chemical fields, particularly in organic chemistry and in the chemistry of natural products, which is clearly reflected in the early prizes. The Nobel Committee, in addition, showed great openness and foresight by recognizing the other border, that towards biology, already in 1907 with the prize to Eduard Buchner “for his biochemical researches and his discovery of cell-free fermentation”.
1.2 The mechanics of the work in the Nobel committee for chemistry
According to the statutes of the Nobel Foundation, the Nobel Committees should have five members, but the Committee for Chemistry has in recent decades chosen to widen its expertise by adding a number of adjunct members (five in 1998) with the same voting rights as the regular members. Until recently there was no limit other than age on how many times regular members could be re-elected for 3-year terms, so that some members sat on the Committee for a very long period. For example, Professor Arne Westgren of Stockholm, who was secretary of the Nobel Committees for Physics and for Chemistry 1926-1943, was also Chairman of the Committee for Chemistry 1944-1965. Present rules, however, only allow two re-elections, so that a member’s maximum total time on the Committee will be nine years.
Only persons that have been properly nominated before 31 January can be considered for the Nobel Prize in a given year. Consequently, the Nobel Committee starts its work by sending out invitations to nominate in the autumn of the preceding year. Recipients of these invitations, for both Physics and Chemistry, are: 1) Swedish and foreign members of the Royal Swedish Academy of Sciences; 2) members of the Nobel Committees for Physics and for Chemistry; 3) Nobel Laureates in Physics and Chemistry; 4) professors in Physics and Chemistry in Scandinavian universities and at Karolinska Institutet; 5) professors in these subjects in a number of universities outside Scandinavia, selected on a rotation basis by the Academy of Sciences; and 6) other scientists that the Academy chooses to invite.
In the initial years of the Nobel Prize, about 300 invitations to nominate for the Nobel Prize for Chemistry were sent out, but this number has increased over the years and was as high as 2,650 in 1998. The number of nominations received has also increased dramatically from 20-40 during the first decade to 400-500 in the 1990s. The number of candidates is usually smaller than the number of nominations, since many candidates receive more than one nomination. During the first few years only about 10 scientists were nominated, but in recent years this number has been in the range of 250-350.
The invitations to nominate are personal, and it is stressed that nominations should not be discussed with the candidate or with colleagues. This is unfortunately not always respected as is obvious from the fact that many identically worded nominations are some years received from the same university. For this reason the Committee does not put much weight on the number of nominations a given candidate receives, unless clearly independent nominations come from different universities in different countries. This attitude was not taken in earlier years however, as is evident from the following statement made by Committee Chairman Arne Westgren, in a survey over the first 60 years of the Nobel Prize for Chemistry [1]: “In fact, if a scientist is proposed by a large number of sponsors in the preliminary international voting, he is normally selected by the Academy.”
Often the same candidate receives nominations both for chemistry and for physics or for chemistry and for medicine. This problem was met already in 1903, when Arrhenius had been nominated both for the Prize for Chemistry and that for Physics, and in its deliberations the Committee for Chemistry suggested that he should be awarded half of each Prize, but this idea was rejected by the Committee for Physics. Because of such borderline problems, the Committee for Chemistry nowadays has joint meetings with those for Physics and for Physiology or Medicine. However, as pronounced by Westgren [1]: “It is now generally recognized that the important thing is to decide whether work which can with equal justice be reckoned as chemistry and physics or chemistry and medicine, is in fact worthy of a Nobel Prize.” For example, Peter Mitchell, who received the 1978 Nobel Prize for Chemistry, could with equal justice have been awarded the Prize for Physiology or Medicine.
Nobel’s will laid down that the prize should be awarded for work done during the preceding year, but in the statutes governing the committee work this has been interpreted to mean the most recent results, or for older work provided its significance has only recently been demonstrated. It was undoubtedly this rule that excluded Stanislao Cannizzaro from receiving one of the first Nobel Prizes, since his work on drawing up a reliable table of atomic weights, helping to establish the periodic system, was done in the middle of the 19th century. A more recent example is Henry Eyring, whose brilliant theory for the rates of chemical reactions, published in 1935, was apparently not understood by members of the Nobel Committee until much later. As a compensation the Royal Swedish Academy of Sciences gave him, in 1977, its highest honor, other than the Nobel Prize, the Berzelius Medal in gold.
2. The first decade of Nobel Prizes for Chemistry
So much fundamental work in chemistry had been carried out during the last two decades of the 19th century that, as stated by Westgren [1], “During the first few years the Academy was chiefly faced with merely deciding the order in which these scientists should be awarded the prize.” For the first prize in 1901 the Academy had to consider 20 nominations, but no less than 11 of these named van’t Hoff, who was also chosen by the Committee for Chemistry. van’t Hoff had already during his thesis work in Utrecht in 1874 published his suggestion that the carbon atom has its four valences directed towards the corners of a regular tetrahedron, a concept which is the very foundation of modern organic chemistry. The Nobel Prize was, however, awarded for his later work on chemical kinetics and equilibria and on the osmotic pressure in solution, published in 1884 and 1886, when he held a professorship in Amsterdam. When he received the prize he had, however, left this for a position at Akademie der Wissenschaften in Berlin in 1896.
In his 1886 work van’t Hoff showed that most dissolved chemical compounds give an osmotic pressure equal to the gas pressure they would have exerted in the absence of the solvent. An apparent exception was aqueous solutions of electrolytes (acids, bases and their salts), but in the following year Arrhenius showed that this anomaly could be explained, if it is assumed that electrolytes in water dissociate into ions. Arrhenius had already presented the rudiments of his dissociation theory in his doctoral thesis, which was defended in Uppsala in 1884 and was not entirely well received by the faculty. It was, however, strongly supported by Ostwald in Riga, who, in fact, travelled to Uppsala to initiate a collaboration with Arrhenius. In 1886-1890 Arrhenius did work with Ostwald, first in Riga and then in Leipzig, and also with van’t Hoff in Berlin. When Arrhenius was awarded the Nobel Prize for Chemistry in 1903, he was since 1895 professor of physics in Stockholm, and he was also nominated for the Prize for Physics (see Section 1).
The award of the Nobel Prize for Chemistry in 1909 to Ostwald was chiefly in recognition of his work on catalysis and the rates of chemical reactions. Ostwald had in his investigations, following up observations in his thesis in 1878, shown that the rate of acid-catalyzed reactions is proportional to the square of the strength of the acid, as measured by titration with base. His work offered support not only to Arrhenius’ theory of dissociation but also to van’t Hoff’s theory for osmotic pressure. Ostwald was founder and editor of Zeitschrift für Physikalische Chemie, the publication of which is generally regarded as the birth of this new branch of chemistry.
Three of the Nobel Prizes for Chemistry during the first decade were awarded for pioneering work in organic chemistry. In 1902 Emil Fischer, then in Berlin, was given the prize for “his work on sugar and purine syntheses”. Fischer’s work is an example of the growing interest from organic chemists in biologically important substances, thus laying the foundation for the development of biochemistry, and at the time of the award Fischer mainly devoted himself to the study of proteins. Another major influence from organic chemistry was the development of chemical industry, and a chief contributor here was Fischer’s teacher, Adolf von Baeyer in Munich, who was awarded the prize in 1905 “in recognition of his services in the advancement of organic chemistry and the chemical industry, … .” His contributions include, in particular, structure determination of organic dyes (indigo, eosin) and the study of aromatic compounds (terpenes). The third Laureate working in organic chemistry was Otto Wallach in Göttingen, who, like von Baeyer, contributed to alicyclic chemistry, studying not only terpenes but also camphor and other components of ethereal oils. At the award ceremony in 1910 the importance of his discoveries for chemical industry was emphasized.
Two of the early prizes were given for the discovery of new chemical elements. Sir William Ramsay from London received the 1904 Nobel Prize for Chemistry for his discovery of a number of noble gases, a new group of chemically unreactive elements. The first one isolated was argon (“the inactive one”), which Ramsay discovered in 1894, in collaboration with Lord Rayleigh [John William Strutt Rayleigh] of the Royal Institution, who was awarded the Prize for Physics in the same year, his investigations of the density of air and other gases forming the basis for this discovery. The following year Ramsay found helium, observed earlier only in the solar spectrum (hence its name), in emanations from radium, thus anticipating later prizes for nuclear chemistry (see below). Later, in 1898 he also discovered, by fractional distillation of liquid air, neon (“the new one”), krypton (“the hidden one”) and xenon (“the strange one”). The isolation of another element, fluorine, by Henri Moissan in Paris was honored with the 1906 Nobel Prize. In attempts to prepare artificial diamonds Moissan had also developed an electric furnace, and this was specifically mentioned in the prize citation, perhaps a reflection of the stipulation in Nobel’s will that the Prize for Chemistry can be given “for the most important discovery or improvement”.
Ernest Rutherford [Lord Rutherford since 1931], professor of physics in Manchester, was awarded the Nobel Prize for Chemistry in 1908 for his investigations of the chemistry of radioactive substances. The discovery of radioactivity had already been recognized with the Nobel Prize for Physics in 1903, but what Rutherford established was the transformation of one element into another, earlier the alchemist’s dream. In his studies of uranium disintegration he found two types of radiation, named a– and b-rays, and by their deviation in electric and magnetic fields he could show that a-rays consist of positively charged particles. His demonstration that these particles are helium nuclei came in the same year as he received the Nobel Prize. Even if the importance of Rutherford’s work for chemistry is obvious, he naturally had also received many nominations for the Nobel Prize for Physics (see Section 1).
In 1897 Eduard Buchner, at the time professor in Tübingen, published results demonstrating that the fermentation of sugar to alcohol and carbon dioxide can take place in the absence of yeast cells. Earlier it had generally been considered that living cells possess a “vital force”, which makes the life processes possible, even if a few prominent chemists, foremost Jöns Jacob Berzelius and Justus von Liebig, had advocated a chemical basis for life. The vitalistic outlook had been fiercely defended by Louis Pasteur, who maintained that alcoholic fermentation can only occur in the presence of living yeast cells. Buchner’s experiments showed unequivocally that fermentation is a catalytic process caused by the action of enzymes, as had been suggested by Berzelius for all life processes, and Buchner called his extract zymase (“enzymes in yeast”). Because of Buchner’s experiment, 1897 is generally regarded as the birth date for biochemistry proper. Buchner was awarded the Nobel Prize for Chemistry in 1907, when he was professor at the agricultural college in Berlin. This confirmed the prediction of his former teacher, Adolf von Baeyer: “This will make him famous, in spite of the fact that he lacks talent as a chemist.”
3. The Nobel Prizes for Chemistry 1911-2000
A survey of the Nobel Prizes for Chemistry awarded during the 20th century, reveals that the development of this field includes breakthroughs in all of its branches, with a certain dominance for progress in physical chemistry and its subcategories (chemical thermodynamics and chemical change), in chemical structure, in several areas of organic chemistry as well as in biochemistry. Of course, the borders between different areas are diffuse, therefore many Laureates will be mentioned in more than one place.
3.1 General and physical chemistry
The Nobel Prize for Chemistry in 1914 was awarded to Theodore William Richards of Harvard University for “his accurate determinations of the atomic weight of a large number of chemical elements”. Most atomic weights in Cannizzaros table (see Section 1.2) had already been determined in the 19th century, particularly by the Belgian chemist Jean Servais Stas, but Richards showed that many of them were in error, mainly because Stas had worked with very concentrated solutions, leading to co-precipitation. In 1913 Richards had discovered that the atomic weight of natural lead and of that formed in radioactive decay of uranium minerals differ. This pointed to the existence of isotopes, i.e. atoms of the same element with different atomic weights, which was accurately demonstrated by Francis William Aston at Cambridge University, with the aid of an instrument developed by him, the mass spectrograph. Aston also showed that the atomic weights of pure isotopes are, within the resolution of his experiment, integral numbers, with the exception of hydrogen, for which he obtained the atomic weight 1.008. For his achievements Aston received the Nobel Prize for Chemistry in 1922.
One branch of physical chemistry deals with chemical events at the interface of two phases, for example, solid and liquid, and phenomena at such interfaces have important applications all the way from technical to physiological processes. Detailed studies of adsorption on surfaces, were carried out by Irving Langmuir at the research laboratory of General Electric Company, and when he was awarded the Nobel Prize for Chemistry in 1932, he was the first industrial scientist to receive this distinction.
Two of the Prizes for Chemistry in more recent decades have been given for fundamental work in the application of spectroscopic methods to chemical problems. Spectroscopy had already been recognized with Prizes for Physics in 1952, 1955 and 1961, when Gerhard Herzberg, a physicist at the University of Saskatchewan, received the Nobel Prize for Chemistry in 1971 for his molecular spectroscopy studies “of the electronic structure and geometry of molecules, particularly free radicals”. The most used spectroscopic method in chemistry is undoubtedly NMR (nuclear magnetic resonance), and Richard R. Ernst at ETH in Zürich was given the Nobel Prize for Chemistry in 1991 for “the development of the methodology of high resolution nuclear magnetic resonance (NMR) spectroscopy”. Ernst’s methodology has now made it possible to determine the structure in solution (in contrast to crystals; cf. Section 3.5) of large molecules, such as proteins.
3.2 Chemical thermodynamics
The first Nobel Prize for Chemistry, that to van’t Hoff, was in part for work in chemical thermodynamics, and many later contributions in this area have also been recognized with Nobel Prizes. Already in 1920 Walther Hermann Nernst of Berlin received this award for work in thermochemistry, despite a 16-year opposition to this recognition from Arrhenius [2]. Nernst had shown that it is possible to determine the equilibrium constant for a chemical reaction from thermal data, and in so doing he formulated what he himself called the third law of thermodynamics. This states that the entropy, a thermodynamic quantity, which is a measure of the disorder in the system, approaches zero as the temperature goes towards absolute zero. van’t Hoff had derived the mass action equation in 1886, with the aid of the second law which says, that the entropy increases in all spontaneous processes [this had already been done in 1876 by J. Willard Gibbs at Yale, who certainly had deserved a Nobel Prize, but his work had been published in an obscure place]. According to the second law, heat of reaction is not an accurate measure of chemical equilibrium, as had been assumed by earlier investigators. But Nernst showed in 1906 that it is possible with the aid of the third law, to derive the necessary parameters from the temperature dependence of thermochemical quantities.
To prove his heat theorem (the third law) Nernst carried out thermochemical measurements at very low temperatures, and such studies were extended in the 1920s by G.N. Lewis (see Section 1.1) in Berkeley. Lewis’s new formulation of the third law was confirmed by his student William Francis Giauque, who extended the temperature range experimentally accessible by introducing the method of adiabatic demagnetization in 1933. With this he managed to reach temperatures a few thousandths of a degree above absolute zero and could thereby provide extremely accurate entropy estimates. He also showed that it is possible to determine entropies from spectroscopic data. Giauque was awarded the Nobel Prize for Chemistry in 1949 for his contributions to chemical thermodynamics.
The next Nobel Prize given for work in thermodynamics went to Lars Onsager of Yale University in 1968 for contributions to the thermodynamics of irreversible processes. Classical thermodynamics deals with systems at equilibrium, in which the chemical reactions are said to be reversible, but many chemical systems, for example, the most complex of all, living organisms, are far from equilibrium and their reactions are said to be irreversible. With the aid of statistical mechanics Onsager developed in 1931 his so-called reciprocal relations, describing the flow of matter and energy in such systems, but the importance of his work was not recognized until the end of the 1940s. A further step forward in the development of non-equilibrium thermodynamics was taken by Ilya Prigogine in Bruxelles, whose theory of dissipative structures was awarded the Nobel Prize for Chemistry in 1977.
3.3 Chemical change
The chief method to get information about the mechanism of chemical reactions is chemical kinetics, i.e. measurements of the rate of the reaction as a function of reactant concentrations as well as its dependence on temperature, pressure and reaction medium. Important work in this area had been done already in the 1880s by two of the early Laureates, van’t Hoff and Arrhenius, who showed that it is not enough for molecules to collide for a reaction to take place. Only molecules with sufficient kinetic energy in the collision do, in fact, react, and Arrhenius derived an equation in 1889 allowing the calculation of this activation energy from the temperature dependence of the reaction rate. With the advent of quantum mechanics in the 1920s (see Section 3.4), Eyring developed his transition-state theory in 1935 and this showed that the activation entropy is also important. Strangely, Eyring never received a Nobel Prize (see Section 1.2).
In 1956 Sir Cyril Norman Hinshelwood of Oxford and Nikolay Nikolaevich Semenov from Moscow shared the Nobel Prize for Chemistry “for their researches into the mechanism of chemical reactions”. Among Hinshelwood’s major contributions his detailed elucidation of the mechanism for the reaction between oxygen and hydrogen can be mentioned, whereas Semenov’s award was for his studies of so-called chain reactions.
A limit in investigating reaction rates is set by the speed with which the reaction can be initiated. If this is done by rapid mixing of the reactants, the time limit is about one thousandth of a second (millisecond). In the 1950s Manfred Eigen from Göttingen developed chemical relaxation methods that allow measurements in times as short as a thousandth or a millionth of a millisecond (microseconds or nanoseconds). The methods involve disturbing an equilibrium by rapid changes in temperature or pressure and then follow the passage to a new equilibrium. Another way to initiate some reactions rapidly is flash photolysis, i.e. by short light flashes, a method developed by Ronald G.W. Norrish at Cambridge and George Porter (Lord Porter since 1990) in London. Eigen received one-half and Norrish and Porter shared the other half of the Nobel Prize for Chemistry in 1967. The milli- to picosecond time scales gave important information on chemical reactions. However, it was not until it was possible to generate femtosecond laser pulses (10-15 s) that it became possible to reveal when chemical bonds are broken and formed. Ahmed Zewail (born 1946 in Egypt) at California Institute of Technology received the Nobel Prize for Chemistry in 1999 for his development of “femtochemistry” and in particular for being the first to experimentally demonstrate a transition state during a chemical reaction. His experiments relate back to 1889 when Arrhenius (Nobel Prize, 1903) made the important prediction that there must exist intermediates (transition states) in the transformation from reactants to products. Henry Taube of Stanford University was awarded the Nobel Prize for Chemistry in 1983 “for his work on the mechanism of electron transfer reactions, especially in metal complexes”. Even if Taube’s work was on inorganic reactions, electron transfer is important in many catalytic processes used in industry and also in biological systems, for example, in respiration and photosynthesis. The latest prize for work in chemical kinetics was that to Dudley R. Herschbach at Harvard University, Yuan T. Lee of Berkeley and John C. Polanyi from Toronto in 1986. Herschbach and his student Lee introduced the use of fluxes of molecules with well-defined direction and energy, molecular beams. By crossing two such beams they could study details of the reaction between molecules at extremely short times. Another important method to investigate such reaction details is infrared chemiluminescence, introduced by Polanyi. The emission of infrared radiation from the reaction products gives information on the energy distribution in the molecules.
3.4 Theoretical chemistry and chemical bonding
Quantum mechanics, developed in the 1920s, offered a tool towards a more basic understanding of chemical bonds. In 1927 Walter Heitler and Fritz London showed that it is possible to solve exactly the relevant equations for the hydrogen molecule ion, i.e. two hydrogen nuclei sharing a single electron, and thereby calculate the attractive force between the nuclei. For molecules containing more than three elementary particles, even the hydrogen molecule with Lewis’s two-electron bond (see Section 1.1), the equation can, however, not be solved exactly, so one has to resort to approximate methods. A pioneer in developing such methods was Linus Pauling at California Institute of Technology, who was awarded the Nobel Prize for Chemistry in 1954 “for his research into the nature of the chemical bond …” Pauling’s valence-bond (VB) method is rigorously described in his 1935 book Introduction to Quantum Mechanics (written together with E. Bright Wilson, Jr., at Harvard). A few years later (1939) he published an extensive non-mathematical treatment in The Nature of the Chemical Bond, a book which is one of the most read and influential in the entire history of chemistry. Pauling was not only a theoretician, but he also carried out extensive investigations of chemical structure by X-ray diffraction (see Section 3.5). On the basis of results with small peptides, which are building blocks of proteins, he suggested the a-helix as an important structural element. Pauling was awarded the Nobel Peace Prize for 1962, and he is the only person to date to have won two unshared Nobel Prizes.
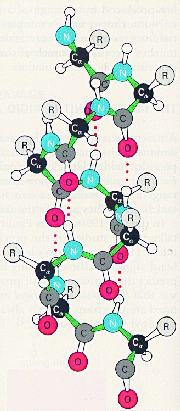
Pauling’s ![]() -helix
-helix
![]() -carbon atoms are black, other carbon atoms grey, nitrogen atoms blue, oxygen atoms red and hydrogen atoms white; R designates amino-acid side chains. The dotted red lines are hydrogen bonds between amide and carbonyl groups in the peptide bonds.
-carbon atoms are black, other carbon atoms grey, nitrogen atoms blue, oxygen atoms red and hydrogen atoms white; R designates amino-acid side chains. The dotted red lines are hydrogen bonds between amide and carbonyl groups in the peptide bonds.
Pauling’s VB method cannot give an adequate description of chemical bonding in many complicated molecules, and a more comprehensive treatment, the molecular-orbital (MO) method, was introduced already in 1927 by Robert S. Mulliken from Chicago and later developed further by him as well as by many other investigators. MO theory considers, in quantum-mechanical terms, the interaction between all atomic nuclei and electrons in a molecule. Mulliken also showed that a combination of MO calculations with experimental (spectroscopic) results provides a powerful tool for describing bonding in large molecules. Mulliken received the Nobel Prize for Chemistry in 1966.
Theoretical chemistry has also contributed significantly to our understanding of chemical reaction mechanisms. In 1981 the Nobel Prize for Chemistry was shared between Kenichi Fukui in Kyoto and Roald Hoffmann of Cornell University “for their theories, developed independently, concerning the course of chemical reactions”. Fukui introduced in 1952 the frontier-orbital theory, according to which the occupied MO with the highest energy and the unoccupied one with the lowest energy have a dominant influence on the reactivity of a molecule. Hoffmann formulated in 1965, together with Robert B. Woodward (see Section 3.8), rules based on the conservation of orbital symmetry, for the reactivity and stereochemistry in chemical reactions.
Rudolph A. Marcus published during ten years, starting in 1956, a series of seminal papers on a comprehensive theory for the rates electron-transfer reactions, the experimental study of which had given Taube a Nobel Prize in 1983 (see Section 3.3). Marcus’s theory predicts how the rate varies with the driving force for the reaction, i.e. the difference in energy between reactants and products, and counter to intuition he found that it does not increase continuously, but goes through a maximum, into the Marcus inverted region, which has later been confirmed experimentally. Marcus was awarded the Nobel Prize for Chemistry in 1992.
The latest Nobel Prize for work in theoretical chemistry was given in 1998 to Walter Kohn of Santa Barbara and John A. Pople of Northwestern University (but a British citizen). The prize to Kohn, a theoretical physicist, was based on his development of density-functional theory, which facilitates detailed calculations both of the geometrical structures of complex molecules and of the energy map of chemical reactions. Pople, a mathematician (but now Professor of Chemistry), was awarded “for his development of computational methods in quantum chemistry”. In particular, Pople has designed computer programs based on classical quantum theory as well as on density-functional theory.
3.5 Chemical structure
The most commonly used method to determine the structure of molecules in three dimensions is X-ray crystallography. The diffraction of X-rays was discovered by Max von Laue in 1912, and this gave him the Nobel Prize for Physics in 1914. Its use for the determination of crystal structure was developed by Sir William Bragg and his son, Sir Lawrence Bragg, and they shared the Nobel Prize for Physics in 1915. The first Nobel Prize for Chemistry for the use of X-ray diffraction went to Petrus (Peter) Debye, then of Berlin, in 1936. Debye did not study crystals, however, but gases, which give less distinct diffraction patterns. He also employed electron diffraction and the measurement of dipole moments to get structural information. Dipole moments are found in molecules, in which the positive and negative charge is unevenly distributed (polar molecules).
Many Nobel Prizes have been awarded for the determination of the structure of biological macromolecules (proteins and nucleic acids). Proteins are long chains of amino-acids, as shown by Emil Fischer (see Section 2), and the first step in the determination of their structure is to determine the order (sequence) of these building blocks. An ingenious method for this tedious task was developed by Frederick Sanger of Cambridge, and he reported the amino-acid sequence for a protein, insulin, in 1955. For this achievement he was awarded the Nobel Prize for Chemistry in 1958. Sanger later received part of a second Nobel Prize for Chemistry for a method to determine the nucleotide sequence in nucleic acids (see Section 3.12), and he is the only scientist so far who has won two Nobel Prizes for Chemistry.
The first protein crystal structures were reported by Max Perutz and Sir John Kendrew in 1960, and these two investigators shared the Nobel Prize for Chemistry in 1962. Perutz had started studying the oxygen-carrying blood pigment, hemoglobin, with Sir Lawrence Bragg in Cambridge already in 1937, and ten years later he was joined by Kendrew, who looked at crystals of the related muscle pigment, myoglobin. These proteins are both rich in Pauling’s a-helix (see Section 3.4), and this made it possible to discern the main features of the structures at the relatively low resolution first used. The same year that Perutz and Kendrew won their prize, the Nobel Prize for Physiology or Medicine went to Francis Crick, James Watson and Maurice Wilkins “for their discoveries concerning the molecular structure of nucleic acids … .” Two years later (1964) Dorothy Crowfoot Hodgkin received the Nobel Prize for Chemistry for determining the crystal structures of penicillin and vitamin B12.
Two later Nobel Prizes for Chemistry in the crystallographic field were given for work on structures of relatively small molecules. William N. Lipscomb of Harvard received the prize in 1976 “for his studies on the structures of boranes illuminating problems of chemical bonding”. In 1985 Herbert A. Hauptman of Buffalo and Jerome Karle of Washington, DC, shared the prize for “the development of direct methods for the determination of crystal structures”. Their methods are called direct, because they yield the structure directly from the diffraction data collected, and they have been indispensable in the determination of the structures of a large number of natural products.
Crystallographic electron microscopy was developed by Sir Aaron Klug in Cambridge, who was awarded the Nobel Prize for Chemistry in 1982. With this technique Klug has investigated the structure of large nucleic acid-protein complexes, such as viruses and chromatin, the carrier of the genes in the cell nucleus. Many of the most important life processes are carried out by proteins associated with biological membranes. This is, for example, true of the two key processes in energy metabolism, respiration and photosynthesis. Attempts to prepare crystals of membrane proteins for structural studies were, however, for many years unsuccessful, but in 1982 Hartmut Michel, then at the Max-Planck-Institut in Martinsried, managed to crystallize a photosynthetic reaction center after a painstaking series of experiments. He then proceeded to determine the three-dimensional structure of this protein complex in collaboration with Johann Deisenhofer and Robert Huber, and this was published in 1985. Deisenhofer, Huber and Michel shared the Nobel Prize for Chemistry in 1988. Michel has later also crystallized and determined the structure of the terminal enzyme in respiration, and his two structures have allowed detailed studies of electron transfer (cf. Sections 3.3 and 3.4) and its coupling to proton pumping, key features of the chemiosmotic mechanism for which Peter Mitchell had already received the Nobel Prize for Chemistry in 1978 (see Section 3.12). Functional and structural studies on the enzyme ATP synthase, connected to this proton pumping mechanism, was awarded one-half of the Nobel Prize for Chemistry in 1997, shared between Paul D. Boyer and John Walker (see Section 3.12).
3.6 Inorganic and nuclear chemistry
Much of the progress in inorganic chemistry during the 20th century has been associated with investigations of coordination compounds, i.e., a central metal ion surrounded by a number of coordinating groups, called ligands. In 1893 Alfred Werner in Zürich presented his coordination theory, and in 1905 he summarized his investigations in this new field in a book (Neuere Anschauungen auf dem Gebiete der anorganischen Chemie), which appeared in no less than five editions from 1905-1923. Compounds in which a metal ion binds several other molecules (ligands), for example, ammonia, had earlier been thought to have a linear structure, in accord with a theory advanced by the Swedish chemist Wilhelm Blomstrand in Lund. Werner showed that such a structure is inconsistent with some experimental facts, and he suggested instead that all the ligand molecules are bound directly to the metal ion. Werner was awarded the Nobel Prize for Chemistry in 1913. Taube’s investigations of electron transfer, awarded in 1983 (see Section 3.3), were mainly carried out with coordination compounds, and vitamin B12 as well as the proteins hemoglobin and myoglobin, investigated by the Laureates Hodgkin, Perutz and Kendrew (see Section 3.5), also belong to this category.
Another early prize for work in inorganic chemistry was that to Fritz Haber from Berlin in 1918 “for the synthesis of ammonia from its elements”, i.e., from nitrogen and hydrogen. The importance of this synthesis is above all in its industrial application in the form of the Haber-Bosch method, which had been developed by Carl Bosch as an improvement (cf. Nobel’s will) of Haber’s original procedure. It allows the manufacture of ammonia on a large scale, and the ammonia can then be used for the production of many different nitrogen-containing chemicals. Bosch shared the Nobel Prize for Chemistry with Friedrich Bergius in 1931 (see Section 3.13).
Much inorganic chemistry in the early 1900s was a consequence of the discovery of radioactivity in 1896, for which Henri Becquerel from Paris was awarded the Nobel Prize for Physics in 1903, together with Pierre and Marie Curie. In 1911 Marie Curie received the Nobel Prize for Chemistry for her discovery of the elements radium and polonium and for the isolation of radium and studies of its compounds, and this made her the first investigator to be awarded two Nobel Prizes. The prize in 1921 went to Frederick Soddy of Oxford for his work on the chemistry of radioactive substances and on the origin of isotopes. In 1934 Frédéric Joliot and his wife Irène Joliot-Curie, the daughter of the Curies, discovered artificial radioactivity, i.e., new radioactive elements produced by the bombardment of non-radioactive elements with a-particles or neutrons. They were awarded the Nobel Prize for Chemistry in 1935 for “their synthesis of new radioactive elements”.
Many elements are mixtures of non-radioactive isotopes (see Section 3.1), and in 1934 Harold Urey of Columbia University had been given the Nobel Prize for Chemistry for his isolation of heavy hydrogen (deuterium). Urey had also separated uranium isotopes, and his work was an important basis for the investigations by Otto Hahn from Berlin. In attempts to make transuranium elements, i.e., elements with a higher atomic number than 92 (uranium), by radiating uranium atoms with neutrons, Hahn discovered that one of the products was barium, a lighter element. Lise Meitner, at the time a refugee from Nazism in Sweden, who had earlier worked with Hahn and taken the initiative for the uranium bombardment experiments, provided the explanation, namely, that the uranium atom was cleaved and that barium was one of the products [3]. Hahn was awarded the Nobel Prize for Chemistry in 1944 “for his discovery of the fission of heavy nuclei”, and it can be wondered why Meitner was not included. Hahn’s original intention with his experiments was later achieved by Edwin M. McMillan and Glenn T. Seaborg of Berkeley, who were given the Nobel Prize for Chemistry in 1951 for “discoveries in the chemistry of transuranium elements”.
The use of stable as well as radioactive isotopes have important applications, not only in chemistry, but also in fields as far apart as biology, geology and archeology. In 1943 George de Hevesy from Stockholm received the Nobel Prize for Chemistry for his work on the use of isotopes as tracers, involving studies in inorganic chemistry and geochemistry as well as on the metabolism in living organisms. The prize in 1960 was given to Willard F. Libby of the University of California, Los Angeles (UCLA), for his method to determine the age of various objects (of geological or archeological origin) by measurements of the radioactive isotope carbon-14.
3.7 General organic chemistry
Contributions in organic chemistry have led to more Nobel Prizes for Chemistry than work in any other of the traditional branches of chemistry. Like the first prize in this area, that to Emil Fischer in 1902 (see Section 2), most of them have, however, been awarded for advances in the chemistry of natural products and will be treated separately (Section 3.9). Another large group, preparative organic chemistry, has also been given its own section (Section 3.8), and here only the prizes for more general contributions to organic chemistry will be discussed. In 1969 the Nobel Prize for Chemistry went to Sir Derek H. R. Barton from London, and Odd Hassel from Oslo for developing the concept of conformation, i.e. the spatial arrangement of atoms in molecules, which differ only by the orientation of chemical groups by rotation around a single bond. This stereochemical concept rests on the original suggestion by van’t Hoff of the tetrahedral arrangement of the four valences of the carbon atom (see Section 2), and most organic molecules exist in two or more stable conformations.
The Nobel Prize for Chemistry in 1975 to Sir John Warcup Cornforth of the University of Sussex and Vladimir Prelog of ETH in Zürich was also based on research in stereochemistry. Not only can a compound have more than one geometric form, but chemical reactions can also have specificity in their stereochemistry, thereby forming a product with a particular three-dimensional arrangement of the atoms. This is especially true of reactions in living organisms, and Cornforth has mainly studied enzyme-catalyzed reactions, so his work borders onto biochemistry (Section 3.12). One of Prelog’s main contributions concerns chiral molecules, i.e. molecules that have two forms differing from one another as the right hand does from the left. Stereochemically specific reactions have great practical importance, as many drugs, for example, are active only in one particular geometric form.
Organometallic compounds constitute a group of organic molecules containing one or more carbon-metal bond, and they are thus the organic counterpart to Werner’s inorganic coordination compounds (see Section 3.6). In 1952 Ernst Otto Fischer and Sir Geoffrey Wilkinson independently described a completely new group of organometallic molecules, called sandwich compounds (see figure below). In such compounds a metal ion is bound not to a single carbon atom but is “sandwiched” between two aromatic organic molecules. Fischer and Wilkinson shared the Nobel Prize for Chemistry in 1973.
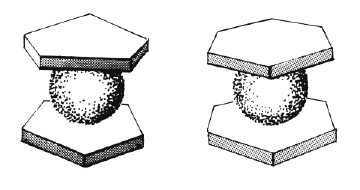 |
| Sandwich compounds |
Work on the interaction of metal ions with organic molecules was also recognized by the prize in 1987, which was shared by Donald J. Cram of UCLA, Jean-Marie Lehn from Strasbourg (and Paris) and Charles J. Pedersen of the Du Pont Company. These three investigators have synthesized molecules with a ring structure, in which the hole in their middle specifically recognizes and binds different metal ions. They can, for example, distinguish between closely related ions, such as those of sodium and potassium, and thus they mimic enzymes in their specificity. The first such compound was synthesized by Pedersen in 1967, and later Lehn and Cram developed increasingly sophisticated organic compounds with cavities and cages in which not only metal ions but other molecules are bound. This research has applications in the whole spectrum of the chemical field, from inorganic chemistry to biochemistry.
George A. Olah from the University of Southern California was awarded the Nobel Prize for Chemistry in 1994 “for his contributions to carbocation chemistry”. Already in the 1920s and 1930s chemists had suggested that positively charged ions of hydrocarbons are formed as short-lived intermediates in organic chemical reactions. Such carbocations were, however, thought to be so reactive and unstable that it would be impossible to prepare them in quantity. Olah’s investigations, starting in the 1960s, contradicted this supposition, since he showed that stable carbocations can be prepared by the use of a new type of extremely acidic compounds (“superacids”), and carbocation chemistry now has a prominent position in all modern textbooks of organic chemistry.
The preparation of a new form of carbon compounds was also recognized by the Nobel Prize for Chemistry in 1996 to Robert F. Curl, Jr., of Rice University, Sir Harold W. Kroto of the University of Sussex and Richard E. Smalley of Rice University. These investigators had in 1985 discovered compounds, called fullerenes, in which 60 or 70 carbon atoms are bound together in clusters in the form of a ball (see figure below). The designation fullerenes is taken from the name of an American architect, R. Buckminster Fuller, who had designed a dome having the form of a football for the 1967 Montreal World Exhibition.
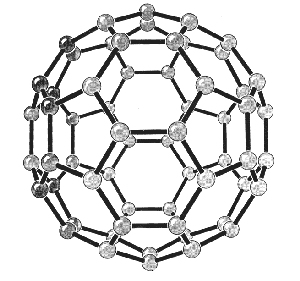
Fullerenes
3.8 Preparative organic chemistry
One of the chief goals of the organic chemist is to be able to synthesize increasingly complex compounds of carbon in combination with various other elements, such as hydrogen, oxygen, nitrogen, sulfur and phosphorus. The first Nobel Prize for Chemistry recognizing pioneering work in preparative organic chemistry was that to Victor Grignard from Nancy and Paul Sabatier from Toulouse in 1912. Grignard had discovered that organic halides can form compounds with magnesium. These compounds, now generally called Grignard reagents, are very reactive, and they are consequently widely used for synthetic purposes. Sabatier was given the prize for developing a method to hydrogenate organic compounds in the presence of metallic catalysts. With his method oils can be converted to saturated fats, and it is, for example, used for margarine production and other industrial processes.
The prize in 1950 was presented to Otto Diels from Kiel and Kurt Alder from Cologne “for their discovery and development of the diene synthesis”, also called the Diels-Alder reaction. In this reaction, which was developed already in 1928, organic compounds containing two double bonds (“dienes”) can effect the syntheses of many cyclic organic substances. During the decades following the original work several industrial applications of the Diels-Alder reaction have been found, for example, in the production of plastics, which may explain the lateness of the prize.
The German organic chemist Hans Fischer from Munich had already done significant work on the structure of hemin, the organic pigment in hemoglobin, when he synthesized it from simpler organic molecules in 1928. He also contributed much to the elucidation of the structure of chlorophyll, and for these important achievements he was awarded the Nobel Prize for Chemistry in 1930 (cf. Section 3.5). He finished his determination of the structure of chlorophyll in 1935, and by the time of his death he had almost completed its synthesis as well.
Robert Burns Woodward from Harvard is rightly considered the founder of the most advanced, modern art of organic synthesis. He designed methods for the total synthesis of a large number of complicated natural products, for example, cholesterol, chlorophyll and vitamin B12. He received the Nobel Prize for Chemistry in 1965, and he would probably have received a second chemistry prize in 1981 for his part in the formulation of the Woodward-Hoffmann rules (see Section 3.4), had it not been for his early death. Work in synthetic organic chemistry was also recognized in 1979 with the prize to Herbert C. Brown of Purdue University and Georg Wittig from Heidelberg, who had developed the use of boron- and phosphorus-containing compounds, respectively, into important reagents in organic synthesis. Another master in chemical synthesis is Elias James Corey from Harvard, who received the prize in 1990. He had made a brilliant analysis of the theory of organic synthesis, which permitted him to synthesize biologically active compounds of a complexity earlier considered impossible.
The Nobel Prize for Chemistry in 1984 was given to Robert Bruce Merrifield of Rockefeller University “for his development of methodology for chemical synthesis on a solid matrix”. Specifically, Merrifield applied this ingenious idea to the synthesis of large peptides and small proteins, for example, ribonuclease (cf. Section 3.12), but the principle has later also been applied to nucleic acid chemistry. In earlier methods each intermediate in the synthesis had to be isolated, which resulted in a drastic drop in yield in syntheses involving a large number of consecutive steps. In Merrifield’s method these isolation steps are replaced by a simple washing procedure, which removes by-products as well as remaining starting materials, and in this way substantial losses are avoided.
3.9 Chemistry of natural product
The synthesis of complex organic molecules must be based on detailed knowledge of their structure. Early work on plant pigments was carried out by Richard Willstätter, a student of Adolf von Baeyer from Munich (see Section 2). Willstätter showed a structural relatedness between chlorophyll and hemin, and he demonstrated that chlorophyll contains magnesium as an integral component. He also carried out pioneering investigations on other plant pigments, such as the carotenoids, and he was awarded the Nobel Prize for Chemistry in 1915 for these achievements. Willstätter’s work laid the ground for the synthetic accomplishments of Hans Fischer (see Section 3.8). In addition, Willstätter contributed to the understanding of enzyme reactions.
The prizes for 1927 and 1928 were both presented to Heinrich Otto Wieland from Munich and Adolf Windaus from Göttingen, respectively, at the Nobel ceremony in 1928. These two chemists had done closely related work on the structure of steroids. The award to Wieland was primarily for his investigations of bile acids, whereas Windaus was recognized mainly for his work on cholesterol and his demonstration of the steroid nature of vitamin D. Wieland had already in 1912, before his prize-winning work, formulated a theory for biological oxidation, according to which removal of hydrogen (dehydrogenation) rather than reaction with oxygen is the dominating process.
Investigations on vitamins were recognized in 1937 and 1938 with the prizes to Sir Norman Haworth from Birmingham and Paul Karrer from Zürich and to Richard Kuhn from Heidelberg. Haworth did outstanding work in carbohydrate chemistry, establishing the ring structure of glucose. He was the first chemist to synthesize vitamin C, and this is the basis for the present large-scale production of this nutrient. Haworth shared the prize with Karrer, who determined the structure of carotene and of vitamin A. Kuhn also worked on carotenoids, and he published the structure of vitamin B2 at the same time as Karrer. He also isolated vitamin B6. In 1939 the Nobel Prize for Chemistry was shared between Adolf Butenandt from Berlin and Leopold Ruzicka (1887-1976) of ETH, Zurich. Butenandt was recognized “for his work on sex hormones”, having isolated estrone, progesterone and androsterone. Ruzicka synthesized androsterone and also testosterone.
The awards for outstanding work in natural-product chemistry continued after World War II. In 1947 Sir Robert Robinson from Oxford received the prize for his studies on plant substances, particularly alkaloids, such as morphine. Robinson also synthesized steroid hormones, and he elucidated the structure of penicillin. Many hormones are of a polypeptide nature, and in 1955 Vincent du Vigneaud of Cornell University was given the prize for his synthesis of two such hormones, vasopressin and oxytocin. Finally, in this area, Alexander R. Todd (Lord Todd since 1962) was recognized in 1957 “for his work on nucleotides and nucleotide co-enzymes”. Todd had synthesized ATP (adenosine triphosphate) and ADP (adenosine diphosphate), the main energy carriers in living cells, and he determined the structure of vitamin B12 (cf. Section 3.5) and of FAD (flavin-adenine dinucleotide).
3.10 Analytical chemistry and separation science
Inorganic chemists, organic chemists and biochemists develop analytical methods as part of their regular research. It is consequently natural that not many Nobel Prizes have been awarded for contributions specifically in analytical chemistry. One such prize was, however, that to Fritz Pregl from Graz in 1923 for his development of organic microanalysis. The medical biochemist from Uppsala, Olof Hammarsten, who gave the presentation speech as Chairman of the Nobel Committee for Chemistry, stressed that Pregl’s work constituted an improvement rather than a discovery, in accord with Nobel’s will. Pregl modified existing methods for quantitative elemental analysis of organic substances to handle very small quantities, which saved time, labor and expense. Another prize in analytical chemistry was given to Jaroslav Heyrovsky from Prague in 1959 for his development of polarographic methods of analysis. In these a dropping mercury electrode is employed to determine current-voltage curves for electrolytes. A given ion reacts at a specific voltage, and the current is a measure of the concentration of this ion.
The analysis of macromolecular constituents in living organisms requires specialized methods of separation. One such method is ultracentrifugation, developed by The Svedberg from Uppsala a few years before he was awarded the Nobel Prize for Chemistry in 1926 “for his work on disperse systems” (see Section 3.11). Svedberg’s student, Arne Tiselius, studied the migration of protein molecules in an electric field, and with this method, named electrophoresis, he demonstrated the complex nature of blood proteins. Tiselius also refined adsorption analysis, a method first used by the Russian botanist, Michail Tswett, for the separation of plant pigments and named chromatography by him. In 1948 Tiselius was given the prize for these achievements. A few years later (1952) Archer J.P. Martin from London and Richard L.M. Synge from Bucksburn (Scotland) shared the prize “for their invention of partition chromatography”, and this method was a major tool in many biochemical investigations later awarded with Nobel Prizes (see Section 3.12).
3.11 Polymers and colloids
Polymeric substances in solution, including life constituents, such as proteins and polysaccharides, are in a colloidal state, i.e., they exist as suspensions of particles one-millionth to one-thousandth of a centimeter in size. In the case of the biological polymers the individual molecules are so large that they form a colloidal suspension, but many other substances can be obtained in a colloidal state. A much-studied example is aggregates of gold atoms, and the Nobel Prize for Chemistry for 1925 was given to Richard Zsigmondy from Göttingen for demonstrating the heterogeneous nature of such gold sols. He did this with the aid of an instrument, the ultramicroscope, which he had developed in collaboration with scientists at the Zeiss factory in Jena. With this instrument the particles and their motion can be observed by the light they scatter at a right angle to the direction of the illuminating light beam. Early work in colloid chemistry had also been carried out by Wolfgang Ostwald, son of the 1909 Laureate Wilhelm Ostwald, but this was not of a caliber earning him a Nobel Prize.
The Svedberg who received the Nobel Prize for Chemistry in 1926, also investigated gold sols. He used Zsigmond’s ultramicroscope to study the Brownian movement of colloidal particles, so named after the Scottish botanist Robert Brown, and confirmed a theory developed by Albert Einstein in 1905 and, independently, by M. Smoluchowski. His greatest achievement was, however, the construction of the ultracentrifuge, with which he studied not only the particle size distribution in gold sols but also determined the molecular weight of proteins, for example, hemoglobin. In the same year as Svedberg got the prize the Nobel Prize for Physics was awarded to Jean Baptiste Perrin of Sorbonne for developing equilibrium sedimentation in colloidal solutions, a method which Svedberg later perfected in his ultracentrifuge. Svedberg’s investigations with the ultracentrifuge and Tiselius’s electrophoresis studies (see Section 3.10) were instrumental in establishing that protein molecules have a unique size and structure, and this was a prerequisite for Sanger’s determination of their amino-acid sequence and the crystallographic work of Kendrew and Perutz (see Section 3.5).
In the 1920s Hermann Staudinger from Freiburg developed the concept of macromolecules. He synthesized many polymers, and he showed that they are long chain molecules. The large plastic industry is largely based on Staudinger’s work. In 1953 he received the Nobel Prize for Chemistry “for his discoveries in the field of macromolecular chemistry”. The prize in 1963 was shared by Karl Ziegler of the Max-Planck-Institute in Mülheim and Giulio Natta from Milan for their discoveries in polymer chemistry and technology. Ziegler demonstrated that certain organometallic compounds (see Section 3.7) can be used to effect polymerization reactions, and Natta showed that Ziegler catalysts can produce polymers with a highly regular three-dimensional structure. Another Nobel Prize for contributions in polymer chemistry was given to Paul J. Flory of Stanford in 1974. Flory carried out fundamental theoretical as well as experimental investigations of the physical chemistry of macromolecules, but his work also led to such important polymers as nylon and synthetic rubber. In 1977 a paper entitled “Synthesis of electrically conducting organic polymers: Halogen derivates of polyacetylene” was published in the Journal of the American Chemical Society, Chemical Communications. The authors of this paper, Alan J. Heeger of the University of California at Santa Barbara, Alan G. MacDiarmid of the University of Pennsylvania and Hideki Shirakawa of the University of Tsukuba, Japan were awarded the Nobel Prize for Chemistry in 2000 for this discovery. The conducting polymers have already given rise to a number of applications such as photodiodes and light-emitting diodes and have future potential to generate microelectronics based upon plastic materials.
3.12 Biochemistry
The second Nobel Prize for discoveries in biochemistry came in 1929, when Sir Arthur Harden from London and Hans von Euler-Chelpin from Stockholm shared the prize for investigations of sugar fermentation, which formed a direct continuation of Buchner’s work awarded in 1907. With his young co-worker, William John Young, Harden had shown in 1906 that fermentation requires a dialysable substance, called co-zymase, which is not destroyed by heat. Harden and Young also demonstrated that the process stops before all sugar (glucose) has been used up, but it starts again on addition of inorganic phosphate, and they suggested that hexose phosphates are formed in the early steps of fermentation. von Euler had done important work on the structure of co-zymase, shown to be nicotinamide adenine dinucleotide (NAD, earlier called DPN). As the number of Laureates can be three, it may seem appropriate for Young to have been included in the award, but Euler’s discovery was published together with Karl Myrbäck, and the number of Laureates is limited to three.
The next biochemical Nobel Prize was given in 1946 for work in the protein field. James B. Sumner of Cornell University received half the prize “for his discovery that enzymes can be crystallized” and John H. Northrop together with Wendell M. Stanley, both of the Rockefeller Institute, shared the other half “for their preparation of enzymes and virus proteins in a pure form”. Sumner had in 1926 crystalized an enzyme, urease, from jack beans and suggested that the crystals were the pure protein. His claim was, however, greeted with great scepticism, and the crystals were suggested to be inorganic salts with the enzyme adsorbed or occluded. Just a few years after Sumner’s discovery Northrop, however, managed to crystalize three digestive enzymes, pepsin, trypsin and chymotrypsin, and by painstaking experiments shown them to be pure proteins. Stanley started his attempt to purify virus proteins in the 1930s, but not until 1945 did he get virus crystals, and this then made it possible to show that viruses are complexes of protein and nucleic acid. The pioneering studies of these three investigators form the basis for the enormous number of new crystal structures of biological macromolecules, which have been published in the second half of the 20th century (cf. Section 3.5).
Several Nobel Prizes for Chemistry have been awarded for work in photosynthesis and respiration, the two main processes in the energy metabolism of living organisms (cf. Section 3.5). In 1961 Melvin Calvin of Berkeley received the prize for elucidating the carbon dioxide assimilation in plants. With the aid of carbon-14 (cf. Section 3.6) Calvin had shown that carbon dioxide is fixed in a cyclic process involving several enzymes. Peter Mitchell of the Glynn Research Laboratories in England was awarded in 1978 for his formulation of the chemiosmotic theory. According to this theory, electron transfer (cf. Sections 3.3 and 3.4) in the membrane-bound enzyme complexes in both respiration and photosynthesis, is coupled to proton translocation across the membranes, and the electrochemical gradient thus created is used to drive the synthesis of ATP (adenosine triphosphate), the energy storage molecule in all living cells. Paul D. Boyer of UCLA and John C. Walker of the MRC Laboratory in Cambridge shared one-half of the 1997 prize for their elucidation of the mechanism of ATP synthesis; the other half of the prize went to Jens C. Skou in Aarhus for the first discovery of an ion-transporting enzyme. Walker had determined the crystal structure of ATP synthase, and this structure confirmed a mechanism earlier proposed by Boyer, mainly on the basis of isotopic studies.
Luis F. Leloir from Buenos Aires was awarded in 1970 “for the discovery of sugar nucleotides and their role in the biosynthesis of carbohydrates”. In particular, Leloir had elucidated the biosynthesis of glycogen, the chief sugar reserve in animals and many microorganisms. Two years later the prize went with one half to Christian B. Anfinsen of NIH and the other half shared by Stanford Moore and William H. Stein, both from Rockefeller University, for fundamental work in protein chemistry. Anfinsen had shown, with the enzyme ribonuclease, that the information for a protein assuming a specific three-dimensional structure is inherent in its amino-acid sequence, and this discovery was the starting point for studies of the mechanism of protein folding, one of the major areas of present-day biochemical research. Moore and Stein had determined the amino-acid sequence of ribonuclease, but they received the prize for discovering anomalous properties of functional groups in the enzyme’s active site, which is a result of the protein fold.
Naturally a number of Nobel Prizes for Chemistry have been given for work in the nucleic acid field. In 1980 Paul Berg of Stanford received one half of the prize for studies of recombinant DNA, i.e. a molecule containing parts of DNA from different species, and the other half was shared by Walter Gilbert from Harvard and Frederick Sanger (see Section 3.5) for developing methods for the determination of the base sequences of nucleic acids. Berg’s work provides the basis of genetic engineering, which has led to the large biotechnology industry. Base sequence determinations are essential steps in recombinant-DNA technology, which is the rationale for Gilbert and Sanger sharing the prize with Berg. Sidney Altman of Yale and Thomas R. Cech of the University of Colorado shared the prize in 1989 “for their discovery of the catalytic properties of RNA”. The central dogma of molecular biology is: DNA –> RNA –> enzyme. The discovery that not only enzymes but also RNA possesses catalytic properties have led to new ideas about the origin of life. The 1993 prize was shared by Kary B. Mullis from La Jolla and Michael Smith from Vancouver, who both have given important contributions to DNA technology. Mullis developed the PCR (“polymerase chain reaction”) technique, which makes it possible to replicate millions of times a specific DNA segment in a complicated genetic material. Smith’s work forms the basis for site-directed mutagenesis, a technique by which it is possible to change a specific amino-acid in a protein and thereby illuminate its functional role.
3.13 Applied chemistry
A few Nobel Prizes for Chemistry have recognized contributions outside the conventional basic chemical fields. The prize in 1931 went to Carl Bosch and Friedrich Bergius , both from Heidelberg, “for the invention and development of chemical high pressure methods”. Bosch had modified Haber’s method for ammonia synthesis (see Section 3.6) to make it suitable for large-scale industrial use. Bergius used high-pressure methods to prepare oil by the hydrogenation of coal, and Bosch, like Bergius working at the large concern I. G. Farben, later improved the procedure by finding a good catalyst for the Bergius process.
Work in agricultural and nutritional chemistry led to the award of Artturi Ilmari Virtanen from Helsinki in 1945. The citation particularly stressed his development of the AIV method, so named after the inventor’s initials. Virtanen had first carried out biochemical studies of nitrogen fixation by plants with the aim of producing protein-rich crops. He then found that the fodder could be preserved with the aid of a mixture of sulfuric and nitric acid (AIV acid).
Finally, basic work in atmospheric and environmental chemistry was recognized in 1995 with the prize to Paul Crutzen, from the Netherlands, working at Stockholm University and later at the Max-Planck-Institute in Mainz, Mario Molina of MIT and F. Sherwood Rowland of UC, Irvine. These three investigators have studied in detail the chemical processes leading to the formation and decomposition of ozone in the atmosphere. In particular, they have shown that the atmospheric ozone layer is very sensitive to emission chemicals produced by human activity, and these discoveries have led to international legislation.
4. Concluding remarks
The first hundred years of Nobel Prizes for Chemistry give a beautiful picture of the development of modern chemistry. The prizes cover the whole spectrum of the basic chemical sciences, from theoretical chemistry to biochemistry, and also a number of contributions to applied chemistry. From a quantitative point of view, organic chemistry dominates with no less than 25 awards. This is not surprising, since the special valence properties of carbon result in an almost infinite variation in the structure of organic compounds. Also, a large number of the prizes in organic chemistry were given for investigations of the chemistry of natural products of increasing complexity and thus are on the border to biochemistry.
As many as 11 prizes have been awarded for biochemical discoveries. Even if the first biochemical prize was already given in 1907 (Buchner), only three awards in this area came in the first half of the century, illustrating the explosive growth of biochemistry in recent decades (8 prizes in 1970-1997). At the other end of the chemical spectrum, physical chemistry, including chemical thermodynamics and kinetics, dominates with 14 prizes, but there has also been 6 prizes in theoretical chemistry. Chemical structure is another large area with 8 prizes, including awards for methodological developments as well as for the determination of the structure of large biological molecules or molecular complexes. Industrial chemistry was first recognized in 1931 (Bergius, Bosch), but many more recent prizes for basic contributions lie close to industrial applications, for example, those in polymer chemistry.
Science is a truly international undertaking, but the western dominance of the Nobel scene is striking. No less than 49 scientists in the United States have received the Nobel Prize for Chemistry, but the majority have been given the prize after World War II. The first US prize was awarded in 1915 (for 1914, Richards), and only two more Americans got the prize before 1946 (Langmuir in 1932, Urey in 1934). German chemists form the second most awarded group with 26 Laureates, but 14 of these received the prize before 1945. Of the 25 British investigators recognized, on the other hand, no less than 19 got the prize in the second half of the century. France has 7 Laureates in chemistry, Sweden and Switzerland 5 each, and the Netherlands and Canada 3. One prize winner each is found in the following countries: Argentina, Austria, Belgium, Czechoslovakia, Denmark, Finland, Italy, Norway and Russia.
Extrapolating the trend of the 20th century Nobel Prizes for Chemistry, it is expected that in the 21st century theoretical and computational chemistry will flourish with the aid of the expansion of computer technology. The study of biological systems may become more dominant and move from individual macromolecules to large interactive systems, for example, in chemical signaling and in neural function, including the brain. And it is to be hoped that the next century will witness a wider national distribution of Laureates.
5. References
Westgren, A., Nobel – The Man and His Prizes, ed. Odelberg, W. (Elsevier, New York, 1972), pp. 279-385.
Kormos Barkan, D., Walther Nernst and the Transition in Modern Physical Science, (Cambridge University Press, 1999).
Rife, P., Lise Meitner and the Dawn of the Nuclear Age, (Birkhäuser, 1999).
* This article was published as a chapter of the book: “The Nobel Prize: The First 100 Years”, Agneta Wallin Levinovitz and Nils Ringertz, eds., Imperial College Press and World Scientific Publishing Co. Pte. Ltd., 2001.
Bo G. Malmström (b. 1927, d. 2000) was Professor of Biochemistry at Göteborg University in 1963-1993. Apart from his doctorate from Uppsala University, he held honorary doctorates from Muhlenberg College in the US and from Utrecht University. He was Visiting Professor at University of Southern California, University of California, Berkeley, Utrecht University and California Institute of Technology, and he also carried out research at Universitá di Roma and Universitá di Firenze. He published over 200 papers, particularly in inorganic biochemistry and bioenergetics. His main interest in later years was the terminal enzyme of respiration, cytochrome oxidase, and its function as a proton pump. In 1996 he started investigating protein folding of redox-active metalloproteins, especially copper proteins. He was a member of the Nobel Committee for Chemistry in 1972-1988 and its chairman in 1977-1988.
Bertil Andersson (b. 1948) is professor in Biochemistry and President of Linköping University, Sweden (1999-2003). He was head of the Dept. of Biochemistry (1987-1995), Dean of the Faculty of Chemical Sciences and prodean of the Science Faculty, (1996-1999) at the University of Stockholm. He has been elected Executive Director of the European Science Foundation for 2004. His honorary appointments include: Member of the Board of the Nobel Foundation, 2000- ; Member (and chairman) of the Nobel Committee for Chemistry 1989-1997, (1997); Member of the Royal Swedish Academy of Sciences, since 1989. He has been President of the chemical section of the Royal Swedish Academy of Sciences, since 1998; Member of Academiae Europea, since 1990; The Finnish Science Society, since 1991; The Australian Academy of Sciences, since 1999; The European Molecular Biology Organization (EMBO), since 1990. He has received Honorary doctorates from Turku University, 2000 and Umeå University, 2002. He has been a Visiting Professor at Imperial College, London since 1990. Prof. Andersson has published a total of 275 papers in photosynthesis research, biological membranes, protein and membrane purification, and light stress.